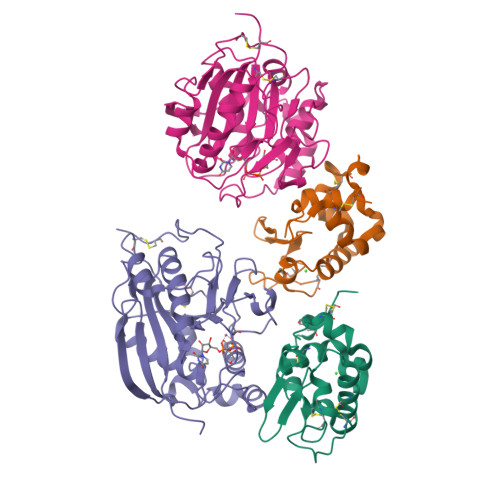
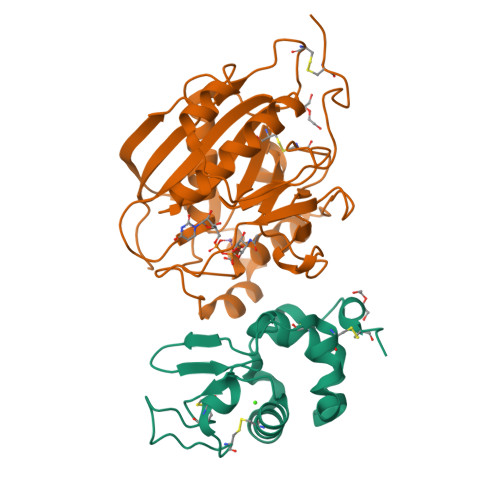
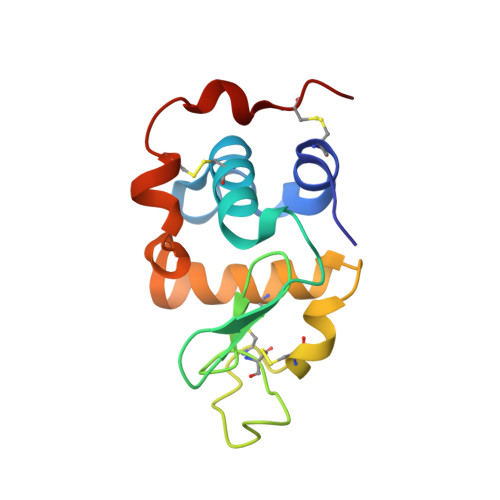
Structure-based design of beta 1,4-galactosyltransferase I (beta 4Gal-T1) with equally efficient N-acetylgalactosaminyltransferase activity: point mutation broadens beta 4Gal-T1 donor specificity.
Ramakrishnan, B., Qasba, P.K.(2002) J Biological Chem 277: 20833-20839
- PubMed: 11916963
- DOI: https://doi.org/10.1074/jbc.M111183200
- Primary Citation of Related Structures:
1OQM - PubMed Abstract:
beta1,4-Galactosyltransferase I (Gal-T1) normally transfers Gal from UDP-Gal to GlcNAc in the presence of Mn(2+) ion. In the presence of alpha-lactalbumin (LA), the Gal acceptor specificity is altered from GlcNAc to Glc. Gal-T1 also transfers GalNAc from UDP-GalNAc to GlcNAc, but with only approximately 0.1% of Gal-T activity. To understand this low GalNAc-transferase activity, we have carried out the crystal structure analysis of the Gal-T1.LA complex with UDP-GalNAc at 2.1-A resolution. The crystal structure reveals that the UDP-GalNAc binding to Gal-T1 is similar to the binding of UDP-Gal to Gal-T1, except for an additional hydrogen bond formed between the N-acetyl group of GalNAc moiety with the Tyr-289 side chain hydroxyl group. Elimination of this additional hydrogen bond by mutating Tyr-289 residue to Leu, Ile, or Asn enhances the GalNAc-transferase activity. Although all three mutants exhibit enhanced GalNAc-transferase activity, the mutant Y289L exhibits GalNAc-transferase activity that is nearly 100% of its Gal-T activity, even while completely retaining its Gal-T activity. The steady state kinetic analyses on the Leu-289 mutant indicate that the K(m) for GlcNAc has increased compared to the wild type. On the other hand, the catalytic constant (k(cat)) in the Gal-T reaction is comparable with the wild type, whereas it is 3-5-fold higher in the GalNAc-T reaction. Interestingly, in the presence of LA, these mutants also transfer GalNAc to Glc instead of to GlcNAc. The present study demonstrates that, in the Gal-T family, the Tyr-289/Phe-289 residue largely determines the sugar donor specificity.
Organizational Affiliation:
Structural Glycobiology Section and Intramural Research Support Program-SAIC, Laboratory of Experimental and Computational Biology, Center for Cancer Research, NCI-Frederick, National Institutes of Health, Frederick, Maryland 21702, USA.