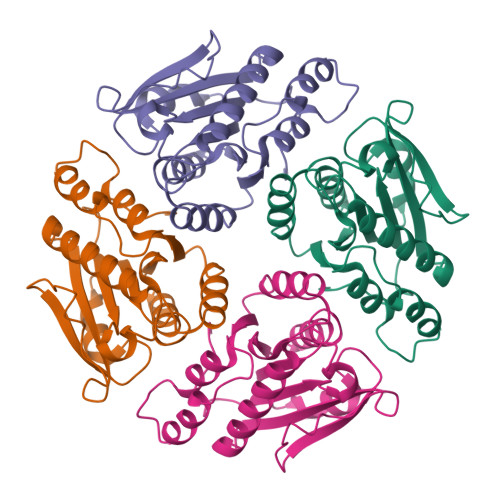
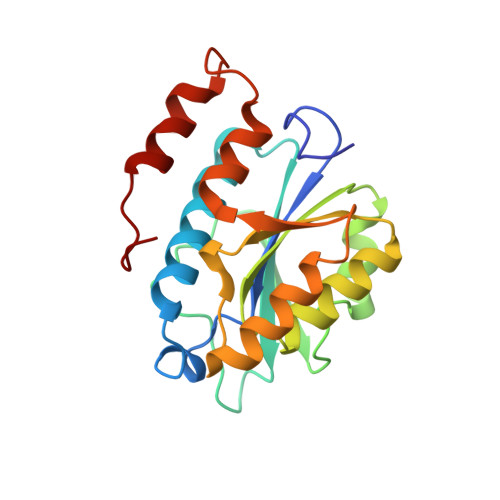
Crystal structures and proposed structural/functional classification of three protozoan proteins from the isochorismatase superfamily.
Caruthers, J., Zucker, F., Worthey, E., Myler, P.J., Buckner, F., Van Voorhuis, W., Mehlin, C., Boni, E., Feist, T., Luft, J., Gulde, S., Lauricella, A., Kaluzhniy, O., Anderson, L., Le Trong, I., Holmes, M.A., Earnest, T., Soltis, M., Hodgson, K.O., Hol, W.G., Merritt, E.A.(2005) Protein Sci 14: 2887-2894
- PubMed: 16199669
- DOI: https://doi.org/10.1110/ps.051783005
- Primary Citation of Related Structures:
1X9G, 1XN4, 1YZV - PubMed Abstract:
We have determined the crystal structures of three homologous proteins from the pathogenic protozoans Leishmania donovani, Leishmania major, and Trypanosoma cruzi. We propose that these proteins represent a new subfamily within the isochorismatase superfamily (CDD classification cd004310). Their overall fold and key active site residues are structurally homologous both to the biochemically well-characterized N-carbamoylsarcosine-amidohydrolase, a cysteine hydrolase, and to the phenazine biosynthesis protein PHZD (isochorismase), an aspartyl hydrolase. All three proteins are annotated as mitochondrial-associated ribonuclease Mar1, based on a previous characterization of the homologous protein from L. tarentolae. This would constitute a new enzymatic activity for this structural superfamily, but this is not strongly supported by the observed structures. In these protozoan proteins, the extended active site is formed by inter-subunit association within a tetramer, which implies a distinct evolutionary history and substrate specificity from the previously characterized members of the isochorismatase superfamily. The characterization of the active site is supported crystallographically by the presence of an unidentified ligand bound at the active site cysteine of the T. cruzi structure.
Organizational Affiliation:
Biomolecular Structure Center M/S 357742, University of Washington, Seattle, WA 98195, USA.