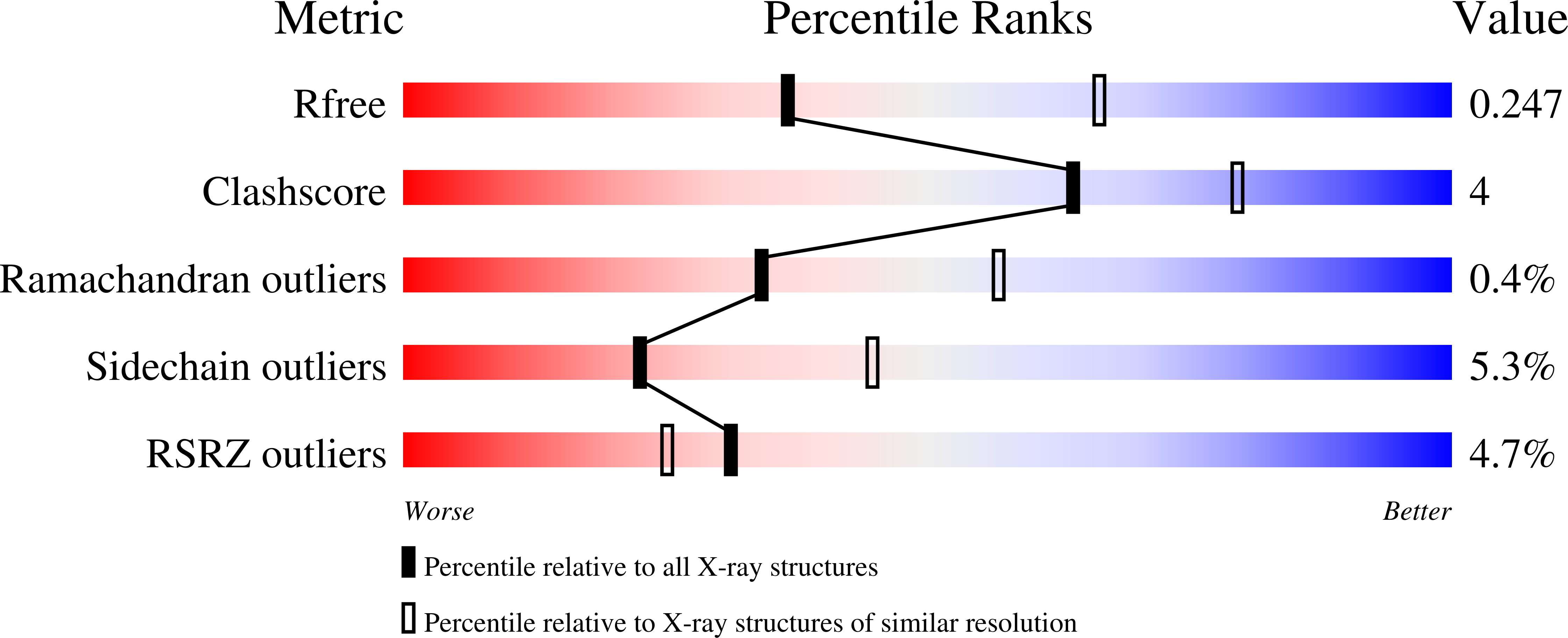
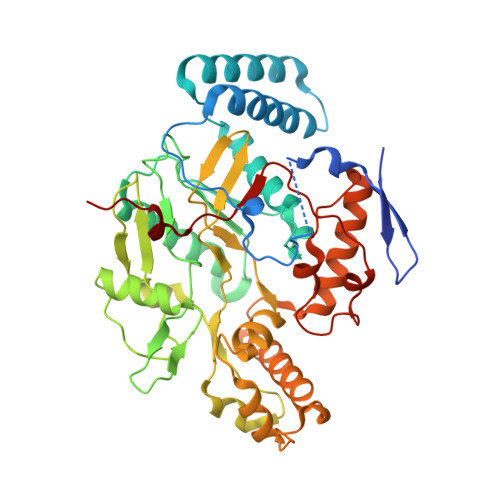
The Discovery of Novel, Potent and Highly Selective Inhibitors of Inducible Nitric Oxide Synthase (Inos).
Cheshire, D.R., Berg, A., Andersson, G.M., Andrews, G., Beaton, H.G., Birkinshaw, T.N., Boughton-Smith, N., Connolly, S., Cook, T.R., Cooper, A., Cooper, S.L., Cox, D., Dixon, J., Gensmantel, N., Hamley, P.J., Harrison, R., Hartopp, P., Kack, H., Leeson, P.D., Luker, T., Mete, A., Millichip, I., Nicholls, D.J., Pimm, A.D., St-Gallay, S.A., Wallace, A.V.(2011) Bioorg Med Chem Lett 21: 2468
- PubMed: 21398123
- DOI: https://doi.org/10.1016/j.bmcl.2011.02.061
- Primary Citation of Related Structures:
2Y37, 4UX6 - PubMed Abstract:
By careful analysis of experimental X-ray ligand crystallographic protein data across several inhibitor series we have discovered a novel, potent and selective series of iNOS inhibitors exemplified by compound 8.
Organizational Affiliation:
Department of Chemistry, AstraZeneca Charnwood, Bakewell Road, Loughborough LE11 5RH, United Kingdom. dave.cheshire@astrazeneca.com