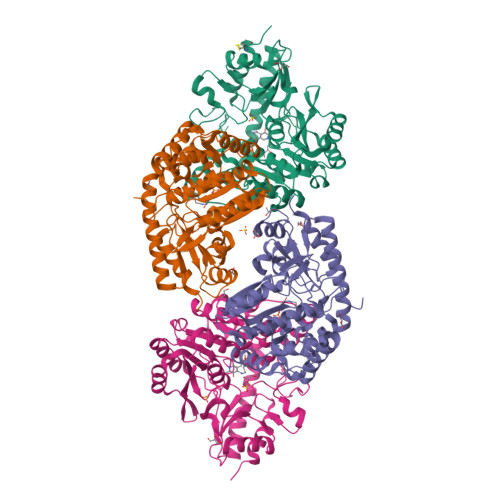
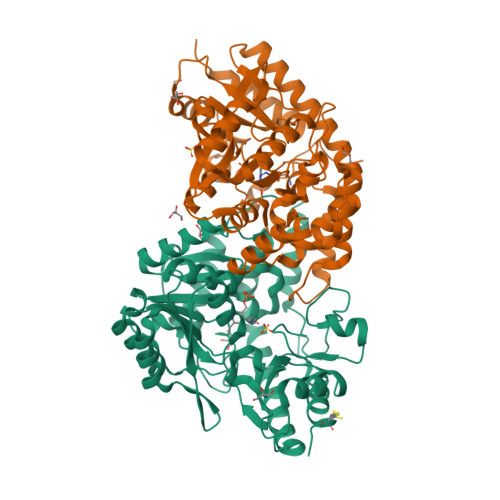
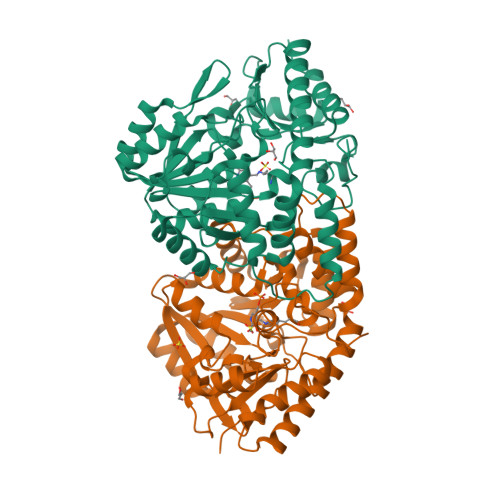
Structural insights into catalysis by beta C-S lyase from Streptococcus anginosus
Kezuka, Y., Yoshida, Y., Nonaka, T.(2012) Proteins 80: 2447-2458
- PubMed: 22674431
- DOI: https://doi.org/10.1002/prot.24129
- Primary Citation of Related Structures:
3B1C, 3B1D, 3B1E - PubMed Abstract:
Hydrogen sulfide (H(2)S) is a causative agent of oral malodor and may play an important role in the pathogenicity of oral bacteria such as Streptococcus anginosus. In this microorganism, H(2)S production is associated with βC-S lyase (Lcd) encoded by lcd gene, which is a pyridoxal 5'-phosphate (PLP)-dependent enzyme that catalyzes the α,β-elimination of sulfur-containing amino acids. When Lcd acts on L-cysteine, H(2)S is produced along with pyruvate and ammonia. To understand the H(2)S-producing mechanism of Lcd in detail, we determined the crystal structures of substrate-free Lcd (internal aldimine form) and two reaction intermediate complexes (external aldimine and α-aminoacrylate forms). The formation of intermediates induced little changes in the overall structure of the enzyme and in the active site residues, with the exception of Lys234, a PLP-binding residue. Structural and mutational analyses highlighted the importance of the active site residues Tyr60, Tyr119, and Arg365. In particular, Tyr119 forms a hydrogen bond with the side chain oxygen atom of L-serine, a substrate analog, in the external aldimine form suggesting its role in the recognition of the sulfur atom of the true substrate (L-cysteine). Tyr119 also plays a role in fixing the PLP cofactor at the proper position during catalysis through binding with its side chain. Finally, we partly modified the catalytic mechanism known for cystalysin, a βC-S lyase from Treponema denticola, and proposed an improved mechanism, which seems to be common to the βC-S lyases from oral bacteria.
Organizational Affiliation:
Department of Structural Biology, School of Pharmacy, Iwate Medical University, Yahaba, Iwate, Japan.