Crystal structure of peptide deformylase from Ehrlichia chaffeensis
Seattle Structural Genomics Center for Infectious Disease (SSGCID), Abendroth, J., Sankaran, B., Edwards, T.E., Staker, B.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Peptide deformylase | 209 | Ehrlichia chaffeensis str. Arkansas | Mutation(s): 0 Gene Names: CDD:29602, def, ECH_0073 EC: 3.5.1.88 | 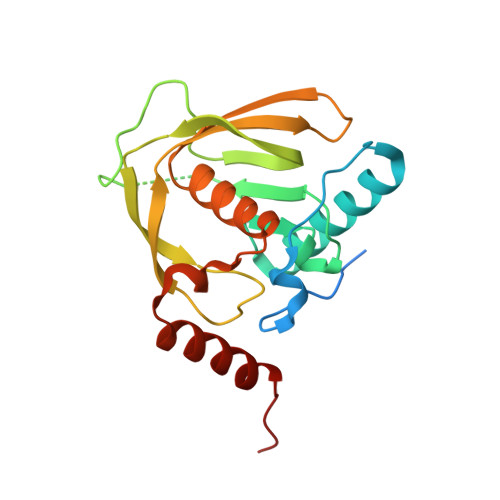 | |
UniProt | |||||
Find proteins for Q2GI30 (Ehrlichia chaffeensis (strain ATCC CRL-10679 / Arkansas)) Explore Q2GI30 Go to UniProtKB: Q2GI30 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q2GI30 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| ZN Query on ZN | C [auth A], F [auth B] | ZINC ION Zn PTFCDOFLOPIGGS-UHFFFAOYSA-N |  | ||
| CL Query on CL | D [auth A], E [auth B] | CHLORIDE ION Cl VEXZGXHMUGYJMC-UHFFFAOYSA-M |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 84.26 | α = 90 |
| b = 64.43 | β = 90 |
| c = 80.91 | γ = 90 |
| Software Name | Purpose |
|---|---|
| BOS | data collection |
| PHASER | phasing |
| REFMAC | refinement |
| XDS | data reduction |
| XSCALE | data scaling |