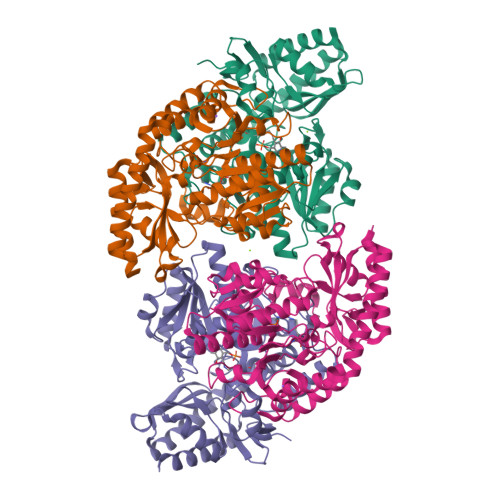
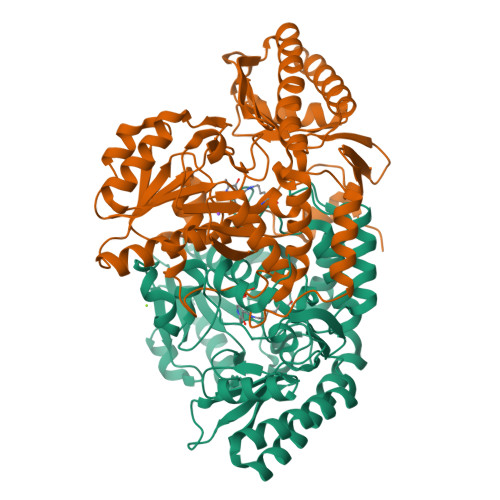
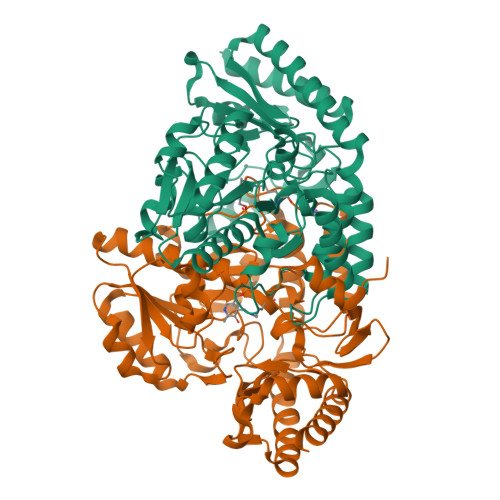
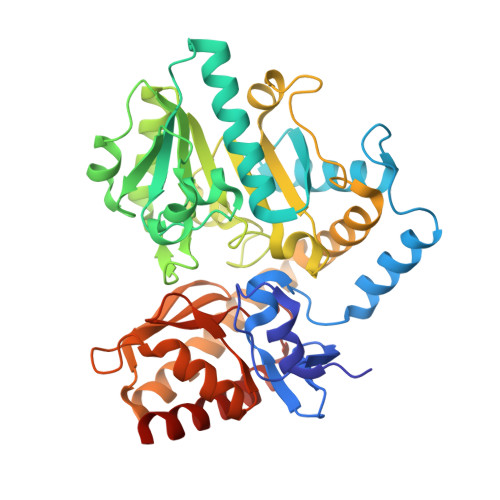
Determination of the Structure of the Catabolic N-Succinylornithine Transaminase (Astc) from Escherichia Coli.
Newman, J., Seabrook, S., Surjadi, R., Williams, C.C., Lucent, D., Wilding, M., Scott, C., Peat, T.S.(2013) PLoS One 8: 58298
- PubMed: 23484010
- DOI: https://doi.org/10.1371/journal.pone.0058298
- Primary Citation of Related Structures:
4ADB, 4ADC, 4ADD, 4ADE - PubMed Abstract:
Escherichia coli possesses two acyl ornithine aminotransferases, one catabolic (AstC) and the other anabolic (ArgD), that participate in L-arginine metabolism. Although only 58% identical, the enzymes have been shown to be functionally interchangeable. Here we have purified AstC and have obtained X-ray crystal structures of apo and holo-AstC and of the enzyme complexed with its physiological substrate, succinylornithine. We compare the structures obtained in this study with those of ArgD from Salmonella typhimurium obtained elsewhere, finding several notable differences. Docking studies were used to explore the docking modes of several substrates (ornithine, succinylornithine and acetylornithine) and the co-substrate glutamate/α-ketogluterate. The docking studies support our observations that AstC has a strong preference for acylated ornithine species over ornithine itself, and suggest that the increase in specificity associated with acylation is caused by steric and desolvation effects rather than specific interactions between the substrate and enzyme.
Organizational Affiliation:
CSIRO Materials, Science and Engineering, Parkville, Australia.