Structural and Kinetic Properties of the Aldehyde Dehydrogenase NahF, a Broad Substrate Specificity Enzyme for Aldehyde Oxidation.
Coitinho, J.B., Pereira, M.S., Costa, D.M., Guimaraes, S.L., Araujo, S.S., Hengge, A.C., Brandao, T.A., Nagem, R.A.(2016) Biochemistry 55: 5453-5463
- PubMed: 27580341
- DOI: https://doi.org/10.1021/acs.biochem.6b00614
- Primary Citation of Related Structures:
4JZ6 - PubMed Abstract:
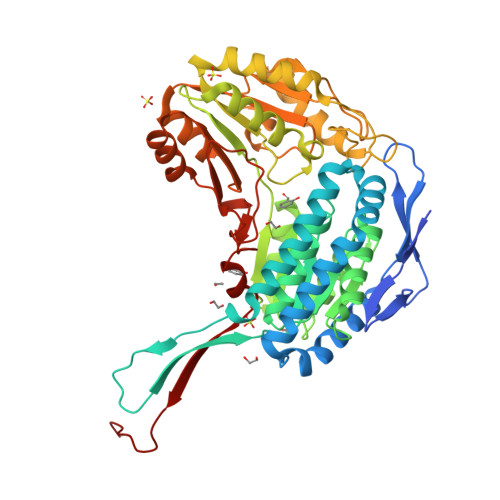
The salicylaldehyde dehydrogenase (NahF) catalyzes the oxidation of salicylaldehyde to salicylate using NAD(+) as a cofactor, the last reaction of the upper degradation pathway of naphthalene in Pseudomonas putida G7. The naphthalene is an abundant and toxic compound in oil and has been used as a model for bioremediation studies. The steady-state kinetic parameters for oxidation of aliphatic or aromatic aldehydes catalyzed by 6xHis-NahF are presented. The 6xHis-NahF catalyzes the oxidation of aromatic aldehydes with large kcat/Km values close to 10(6) M(-1) s(-1). The active site of NahF is highly hydrophobic, and the enzyme shows higher specificity for less polar substrates than for polar substrates, e.g., acetaldehyde. The enzyme shows α/β folding with three well-defined domains: the oligomerization domain, which is responsible for the interlacement between the two monomers; the Rossmann-like fold domain, essential for nucleotide binding; and the catalytic domain. A salicylaldehyde molecule was observed in a deep pocket in the crystal structure of NahF where the catalytic C284 and E250 are present. Moreover, the residues G150, R157, W96, F99, F274, F279, and Y446 were thought to be important for catalysis and specificity for aromatic aldehydes. Understanding the molecular features responsible for NahF activity allows for comparisons with other aldehyde dehydrogenases and, together with structural information, provides the information needed for future mutational studies aimed to enhance its stability and specificity and further its use in biotechnological processes.
Organizational Affiliation:
Departamento de Bioquímica e Imunologia, Instituto de Ciências Biológicas, Universidade Federal de Minas Gerais , Belo Horizonte, MG 31270-901, Brazil.