Crystal structure of DAAO variant
Nakano, S., Yasukawa, K., Kawahara, N., Ishitsubo, E., Tokiwa, H., Asano, Y.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| D-amino-acid oxidase | 342 | Sus scrofa | Mutation(s): 2 Gene Names: DAO EC: 1.4.3.3 | 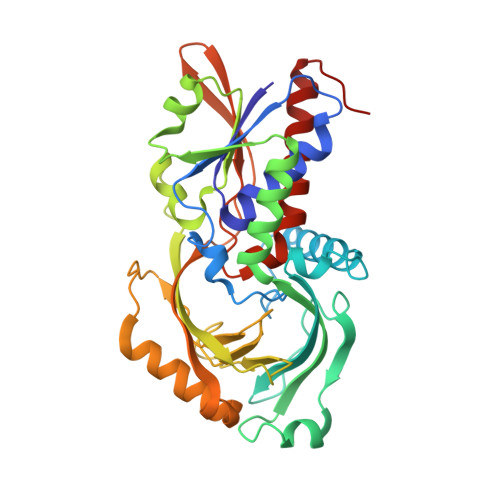 | |
UniProt | |||||
Find proteins for P00371 (Sus scrofa) Explore P00371 Go to UniProtKB: P00371 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P00371 | ||||
Sequence AnnotationsExpand | |||||
| |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| D-amino-acid oxidase | 340 | Sus scrofa | Mutation(s): 2 Gene Names: DAO EC: 1.4.3.3 | 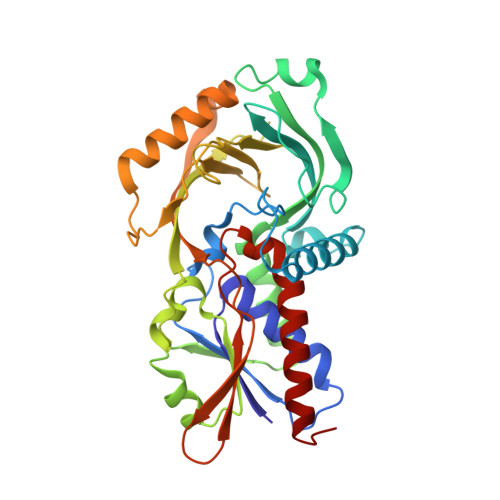 | |
UniProt | |||||
Find proteins for P00371 (Sus scrofa) Explore P00371 Go to UniProtKB: P00371 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P00371 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| FAD Query on FAD | D [auth A], G [auth B] | FLAVIN-ADENINE DINUCLEOTIDE C27 H33 N9 O15 P2 VWWQXMAJTJZDQX-UYBVJOGSSA-N |  | ||
| 96B Query on 96B | C [auth A], F [auth B] | (2R)-2-phenylpyrrolidine C10 H13 N JUTDHSGANMHVIC-SNVBAGLBSA-N |  | ||
| SO4 Query on SO4 | E [auth A], H [auth B], I [auth B] | SULFATE ION O4 S QAOWNCQODCNURD-UHFFFAOYSA-L |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 68.281 | α = 90 |
| b = 91.202 | β = 90 |
| c = 110.007 | γ = 90 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |
| MOLREP | phasing |