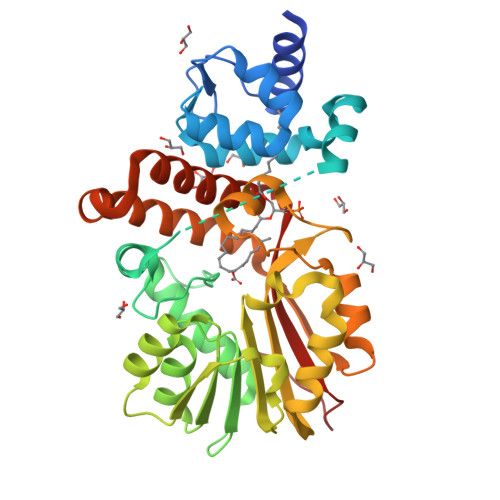
Structural and Functional Analysis of E. coli Cyclopropane Fatty Acid Synthase.
Hari, S.B., Grant, R.A., Sauer, R.T.(2018) Structure 26: 1251
- PubMed: 30057024
- DOI: https://doi.org/10.1016/j.str.2018.06.008
- Primary Citation of Related Structures:
6BQC - PubMed Abstract:
Cell membranes must adapt to different environments. In Gram-negative bacteria, the inner membrane can be remodeled directly by modification of lipids embedded in the bilayer. For example, when Escherichia coli enters stationary phase, cyclopropane fatty acid (CFA) synthase converts most double bonds in unsaturated inner-membrane lipids into cyclopropyl groups. Here we report the crystal structure of E. coli CFA synthase. The enzyme is a dimer in the crystal and in solution, with each subunit containing a smaller N-domain that associates tightly with a larger catalytic C-domain, even following cleavage of the inter-domain linker or co-expression of each individual domain. Efficient catalysis requires dimerization and proper linkage of the two domains. These findings support an avidity-based model in which one subunit of the dimer stabilizes membrane binding, while the other subunit carries out catalysis.
Organizational Affiliation:
Department of Biology, Massachusetts Institute of Technology, Cambridge, MA 02139, USA.