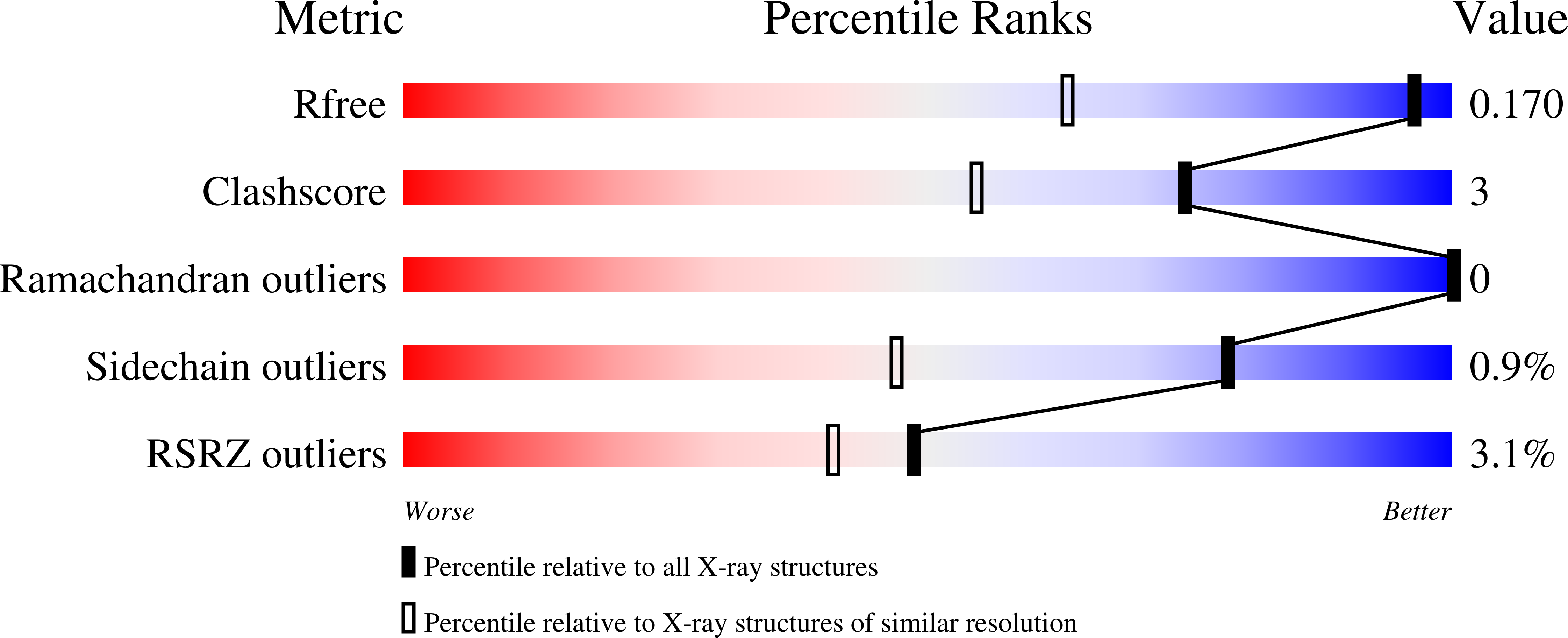
Molecular Basis for Inhibition of Heparanases and beta-Glucuronidases by Siastatin B.
Chen, Y., van den Nieuwendijk, A.M.C.H., Wu, L., Moran, E., Skoulikopoulou, F., van Riet, V., Overkleeft, H.S., Davies, G.J., Armstrong, Z.(2024) J Am Chem Soc 146: 125-133
- PubMed: 38118176
- DOI: https://doi.org/10.1021/jacs.3c04162
- Primary Citation of Related Structures:
8CQI, 8OGX, 8OHQ, 8OHR, 8OHT, 8OHU, 8OHV, 8OHW, 8OHX - PubMed Abstract:
Siastatin B is a potent and effective iminosugar inhibitor of three diverse glycosidase classes, namely, sialidases, β- d -glucuronidases, and N -acetyl-glucosaminidases. The mode of inhibition of glucuronidases, in contrast to sialidases, has long been enigmatic as siastatin B appears too bulky and incorrectly substituted to be accommodated within a β- d -glucuronidase active site pocket. Herein, we show through crystallographic analysis of protein-inhibitor complexes that siastatin B generates both a hemiaminal and a 3-geminal diol iminosugar (3-GDI) that are, rather than the parent compound, directly responsible for enzyme inhibition. The hemiaminal product is the first observation of a natural product that belongs to the noeuromycin class of inhibitors. Additionally, the 3-GDI represents a new and potent class of the iminosugar glycosidase inhibitor. To substantiate our findings, we synthesized both the gluco - and galacto -configured 3-GDIs and characterized their binding both structurally and kinetically to exo-β- d -glucuronidases and the anticancer target human heparanase. This revealed submicromolar inhibition of exo-β- d -glucuronidases and an unprecedented binding mode by this new class of inhibitor. Our results reveal the mechanism by which siastatin B acts as a broad-spectrum glycosidase inhibitor, identify a new class of glycosidase inhibitor, and suggest new functionalities that can be incorporated into future generations of glycosidase inhibitors.
Organizational Affiliation:
Leiden Institute of Chemistry, Leiden University, Einsteinweg 55, 2300 RA Leiden, The Netherlands.