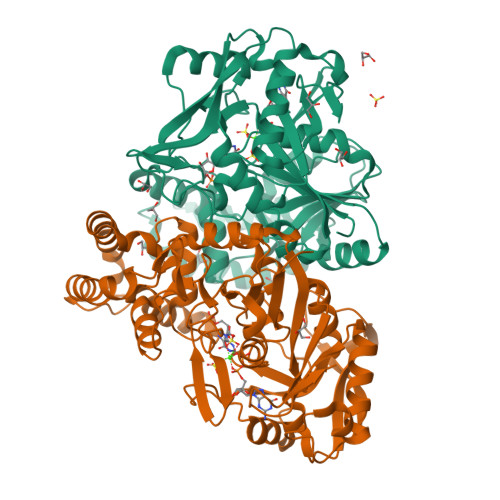
Location Is Everything: Influence of His-Tag Fusion Site on Properties of Adenylosuccinate Synthetase from Helicobacter pylori.
Miskovic, M.Z., Wojtys, M., Winiewska-Szajewska, M., Wielgus-Kutrowska, B., Matkovic, M., Domazet Jurasin, D., Stefanic, Z., Bzowska, A., Lescic Asler, I.(2024) Int J Mol Sci 25
- PubMed: 39062851
- DOI: https://doi.org/10.3390/ijms25147613
- Primary Citation of Related Structures:
9F17 - PubMed Abstract:
The requirement for fast and dependable protein purification methods is constant, either for functional studies of natural proteins or for the production of biotechnological protein products. The original procedure has to be formulated for each individual protein, and this demanding task was significantly simplified by the introduction of affinity tags. Helicobacter pylori adenylosuccinate synthetase (AdSS) is present in solution in a dynamic equilibrium of monomers and biologically active homodimers. The addition of the His 6 -tag on the C-terminus (C-His-AdSS) was proven to have a negligible effect on the characteristics of this enzyme. This paper shows that the same enzyme with the His 6 -tag fused on its N-terminus (N-His-AdSS) has a high tendency to precipitate. Circular dichroism and X-ray diffraction studies do not detect any structural change that could explain this propensity. However, the dynamic light scattering, differential scanning fluorimetry, and analytical ultracentrifugation measurements indicate that the monomer of this construct is prone to aggregation, which shifts the equilibrium towards the insoluble precipitant. In agreement, enzyme kinetics measurements showed reduced enzyme activity, but preserved affinity for the substrates, in comparison with the wild-type and C-His-AdSS. The presented results reinforce the notion that testing the influence of the tag on protein properties should not be overlooked.
Organizational Affiliation:
Department of Chemistry, Faculty of Science, University of Zagreb, Horvatovac 102a, HR-10000 Zagreb, Croatia.