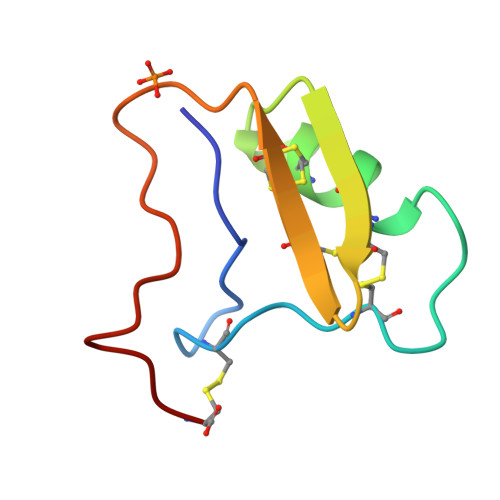
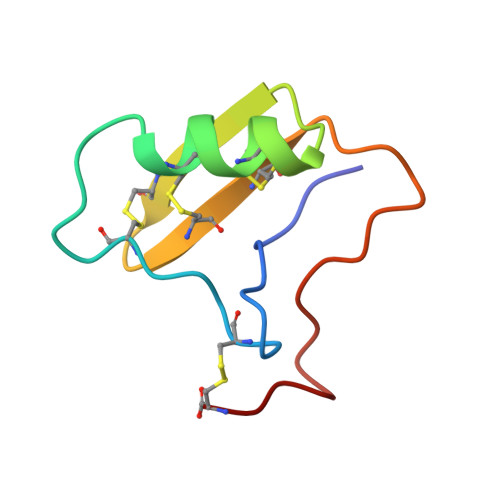
Crystal structure of neurotoxin Ts1 from Tityus serrulatus provides insights into the specificity and toxicity of scorpion toxins.
Polikarpov, I., Junior, M.S., Marangoni, S., Toyama, M.H., Teplyakov, A.(1999) J Mol Biology 290: 175-184
- PubMed: 10388565
- DOI: https://doi.org/10.1006/jmbi.1999.2868
- Primary Citation of Related Structures:
1B7D - PubMed Abstract:
The crystal structure of neurotoxin Ts1, a major component of the venom of the Brazilian scorpion Tityus serrulatus, has been determined at 1.7 A resolution. It is the first X-ray structure of a highly toxic anti-mammalian beta-toxin. The folding of the polypeptide chain of Ts1 is similar to that of other scorpion toxins. A cysteine-stabilised alpha-helix/beta-sheet motif forms the core of the flattened molecule. All residues identified as functionally important by chemical modification and site-directed mutagenesis are located on one side of the molecule, which is therefore considered as the Na+channel recognition site. The distribution of charged and non-polar residues over this surface determines the specificity of the toxin-channel interaction. Comparison to other scorpion toxins shows that positively charged groups at positions 1 and 12 as well as a negative charge at position 2 are likely determinants of the specificity of beta-toxins. In contrast, the contribution of the conserved aromatic cluster to the interaction might be relatively small. Comparison of Ts1 to weak beta-toxins from Centruroides sculpturatus Ewing reveals that a number of basic amino acid residues located on the face of the molecule opposite to the binding surface may account for the high toxicity of Ts1.
Organizational Affiliation:
Laboratório Nacional de Luz Síncrotron (LNLS), Campinas SP, CEP 13083-970, Brazil. igor@lnls.br