Structural basis for specific recognition of single-stranded RNA by Toll-like receptor 13
Song, W., Wang, J., Han, Z., Zhang, Y., Zhang, H., Wang, W., Chang, J., Xia, B., Fan, S., Zhang, D., Wang, J., Wang, H.W., Chai, J.(2015) Nat Struct Mol Biol 22: 782-787
- PubMed: 26323037
- DOI: https://doi.org/10.1038/nsmb.3080
- Primary Citation of Related Structures:
4Z0C - PubMed Abstract:
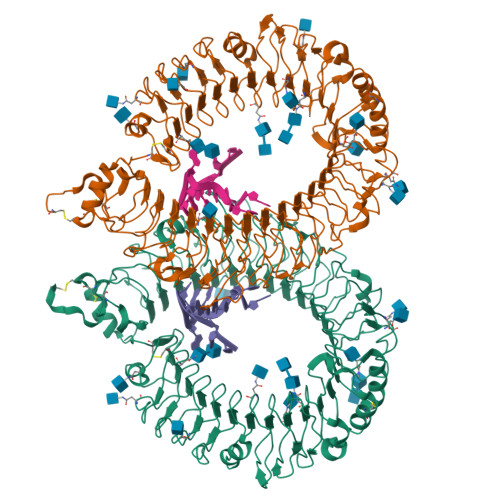
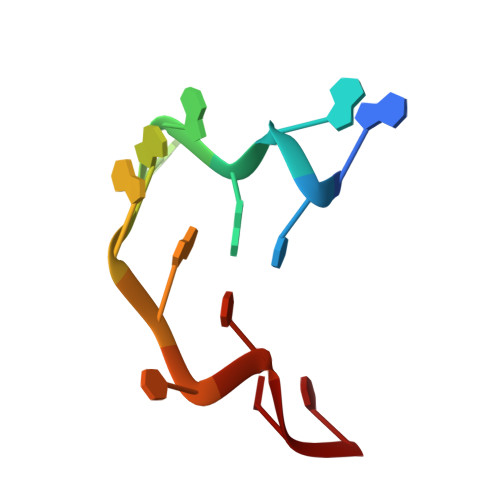
Toll-like receptors (TLRs) have crucial roles in innate immunity, functioning as pattern-recognition receptors. TLR13 recognizes a conserved sequence from bacterial 23S rRNA and then triggers an immune response. Here we report the crystal structure of the mouse TLR13 ectodomain bound by a 13-nt single-stranded (ss) RNA derived from 23S rRNA. The ssRNA induces TLR13 dimerization but assumes a stem-loop-like structure that is completely different from that in the bacterial ribosome but nevertheless is crucial for TLR13 recognition. Most of the RNA nucleotides are splayed out to make base-specific contacts with the concave surface of TLR13, and RNA-specific interactions are important to allow TLR13 to distinguish RNA from DNA. Interestingly, a viral-derived 16-nt ssRNA predicted to form a similar stem-loop-like structure also induces TLR13 activation. Together, our results reveal the structural mechanism of TLR13's sequence- and conformation-specific recognition of ssRNA.
Organizational Affiliation:
Ministry of Education Key Laboratory of Protein Science, Center for Structural Biology, School of Life Sciences, Tsinghua University, Beijing, China.