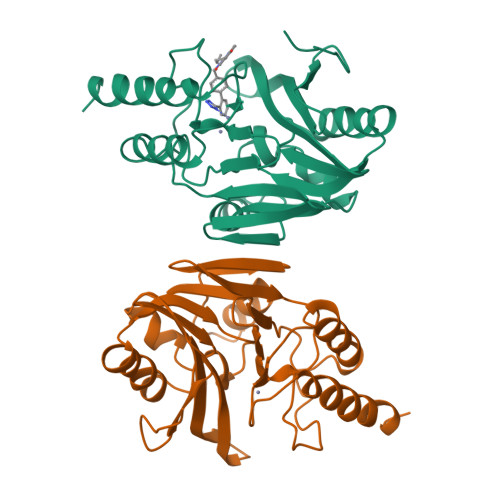
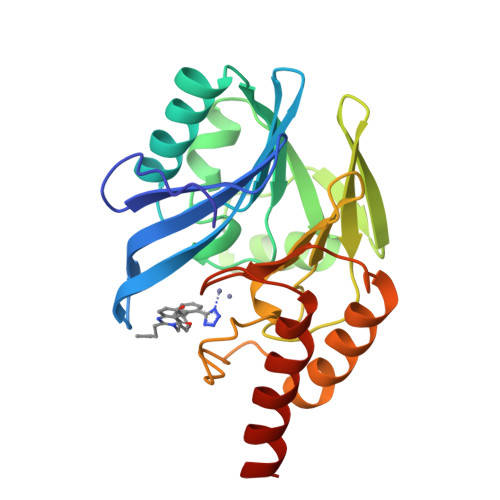
Antibiotic sensitization using biphenyl tetrazoles as potent inhibitors of Bacteroides fragilis metallo-beta-lactamase.
Toney, J.H., Fitzgerald, P.M., Grover-Sharma, N., Olson, S.H., May, W.J., Sundelof, J.G., Vanderwall, D.E., Cleary, K.A., Grant, S.K., Wu, J.K., Kozarich, J.W., Pompliano, D.L., Hammond, G.G.(1998) Chem Biol 5: 185-196
- PubMed: 9545432
- DOI: https://doi.org/10.1016/s1074-5521(98)90632-9
- Primary Citation of Related Structures:
1A8T - PubMed Abstract:
High level resistance to carbapenem antibiotics in gram negative bacteria such as Bacteroides fragilis is caused, in part, by expression of a wide-spectrum metallo-beta-lactamase that hydrolyzes the drug to an inactive form. Co-administration of metallo-beta-lactamase inhibitors to resistant bacteria is expected to restore the antibacterial activity of carbapenems. Biphenyl tetrazoles (BPTs) are a structural class of potent competitive inhibitors of metallo-beta-lactamase identified through screening and predicted using molecular modeling of the enzyme structure. The X-ray crystal structure of the enzyme bound to the BPT L-159,061 shows that the tetrazole moiety of the inhibitor interacts directly with one of the two zinc atoms in the active site, replacing a metal-bound water molecule. Inhibition of metallo-beta-lactamase by BPTs in vitro correlates well with antibiotic sensitization of resistant B. fragilis. BPT inhibitors can sensitize a resistant B. fragilis clinical isolate expressing metallo-beta-lactamase to the antibiotics imipenem or penicillin G but not to rifampicin.
Organizational Affiliation:
Department of Biochemistry, Merck Research Laboratories, P. O. Box 2000, Rahway, NJ 07065-0900, USA. jeff_toney@merck.com