Triiodide Derivatization and Combinatorial Counter-Ion Replacement: Two Methods for Enhancing Phasing Signal Using Laboratory Cu Kalpha X-Ray Equipment
Evans, G., Bricogne, G.(2002) Acta Crystallogr D Biol Crystallogr 58: 976
- PubMed: 12037300
- DOI: https://doi.org/10.1107/s0907444902005486
- Primary Citation of Related Structures:
1GW9, 1GWA, 1GWD, 1GWG - PubMed Abstract:
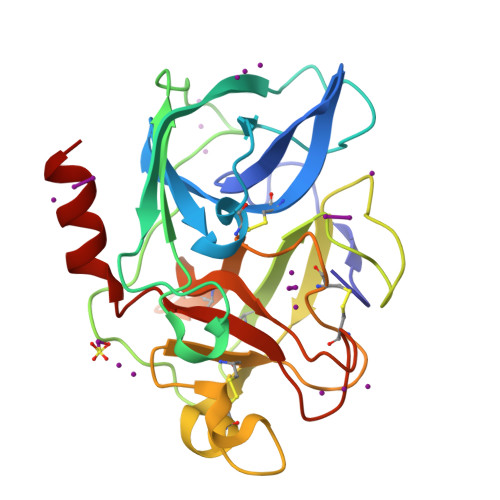
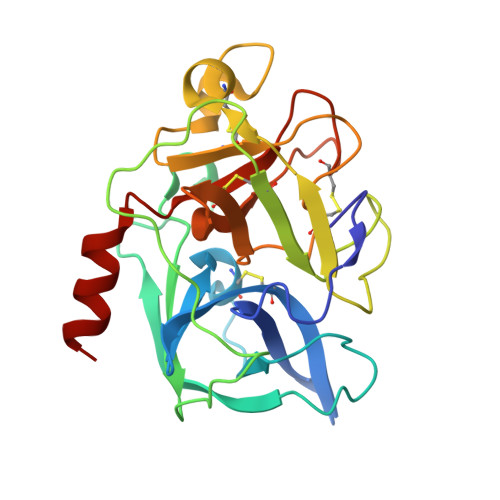
A series of experiments performed at Cu Kalpha wavelength on in-house X-ray equipment are presented which investigate two possibilities for enhancing the experimental phasing signal by means of (i) triiodide/iodide soaks using KI/I(2) and (ii) combinations of counter-ions introduced using the short cryosoak method. Triiodide-derivative crystal structures for five test proteins have been refined and reveal that iodine can bind as polyiodide and single iodide ions through hydrophobic and hydrogen-bonding interactions both at the molecular surface and in intramolecular and intermolecular cavities. In three cases, the structures could be automatically determined with autoSHARP using in-house SAD and SIRAS data. The investigation of combinatorial counter-ion replacement using multiple salts with Na(+) and Cs(+) as cations and I(-) and Cl(-) as anions reveals that, for the case of hen egg-white lysozyme, significant improvement in phasing signal is obtained by the combined use of salts compared with SIRAS methods using native and single short-soak derivative data sets.
Organizational Affiliation:
Global Phasing Limited, Cambridge, England.