A Molecular Switch and Proton-Wire Synchronize the Active Sites in Thiamine-Dependent Enzymes
Frank, R.A.W., Titman, C.M., Pratap, J.V., Luisi, B.F., Perham, R.N.(2004) Science 306: 872
- PubMed: 15514159
- DOI: https://doi.org/10.1126/science.1101030
- Primary Citation of Related Structures:
1W85, 1W88 - PubMed Abstract:
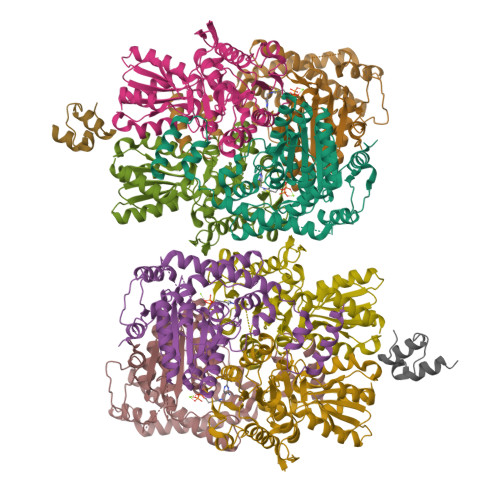
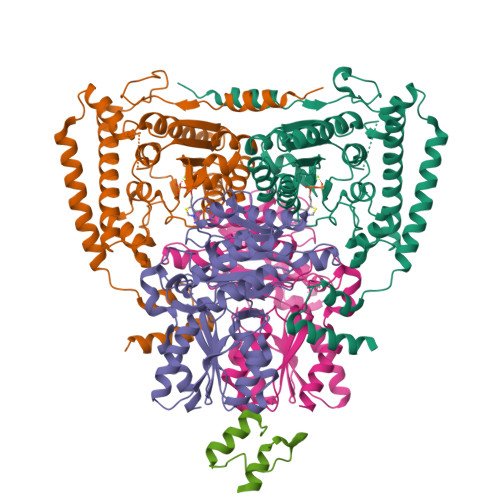
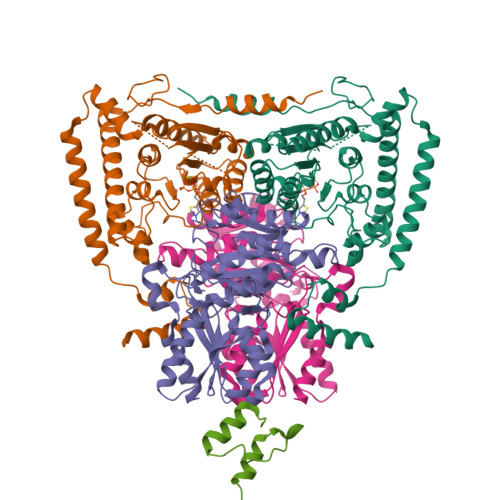
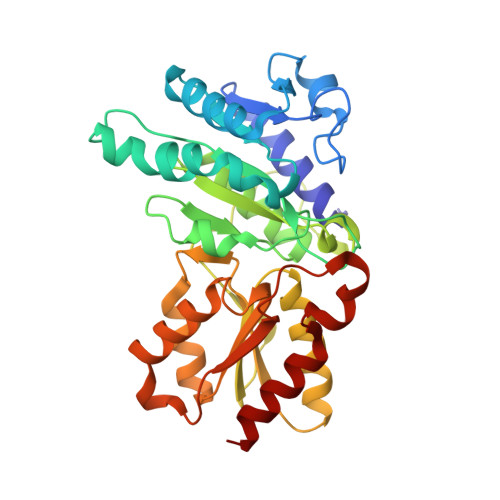
Thiamine diphosphate (ThDP) is used as a cofactor in many key metabolic enzymes. We present evidence that the ThDPs in the two active sites of the E1 (EC 1.2.4.1) component of the pyruvate dehydrogenase complex communicate over a distance of 20 angstroms by reversibly shuttling a proton through an acidic tunnel in the protein. This "proton wire" permits the co-factors to serve reciprocally as general acid/base in catalysis and to switch the conformation of crucial active-site peptide loops. This synchronizes the progression of chemical events and can account for the oligomeric organization, conformational asymmetry, and "ping-pong" kinetic properties of E1 and other thiamine-dependent enzymes.
Organizational Affiliation:
Department of Biochemistry, University of Cambridge, Tennis Court Road, Cambridge, UK.