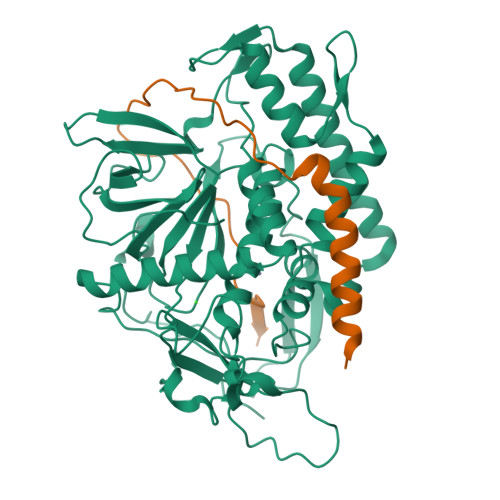
Substrate recognition strategy for botulinum neurotoxin serotype A
Breidenbach, M.A., Brunger, A.T.(2004) Nature 432: 925-929
- PubMed: 15592454
- DOI: https://doi.org/10.1038/nature03123
- Primary Citation of Related Structures:
1XTF, 1XTG - PubMed Abstract:
Clostridal neurotoxins (CNTs) are the causative agents of the neuroparalytic diseases botulism and tetanus. CNTs impair neuronal exocytosis through specific proteolysis of essential proteins called SNAREs. SNARE assembly into a low-energy ternary complex is believed to catalyse membrane fusion, precipitating neurotransmitter release; this process is attenuated in response to SNARE proteolysis. Site-specific SNARE hydrolysis is catalysed by the CNT light chains, a unique group of zinc-dependent endopeptidases. The means by which a CNT properly identifies and cleaves its target SNARE has been a subject of much speculation; it is thought to use one or more regions of enzyme-substrate interaction remote from the active site (exosites). Here we report the first structure of a CNT endopeptidase in complex with its target SNARE at a resolution of 2.1 A: botulinum neurotoxin serotype A (BoNT/A) protease bound to human SNAP-25. The structure, together with enzyme kinetic data, reveals an array of exosites that determine substrate specificity. Substrate orientation is similar to that of the general zinc-dependent metalloprotease thermolysin. We observe significant structural changes near the toxin's catalytic pocket upon substrate binding, probably serving to render the protease competent for catalysis. The novel structures of the substrate-recognition exosites could be used for designing inhibitors specific to BoNT/A.
Organizational Affiliation:
Department of Molecular and Cellular Physiology, Stanford University, Stanford, California 94305, USA.