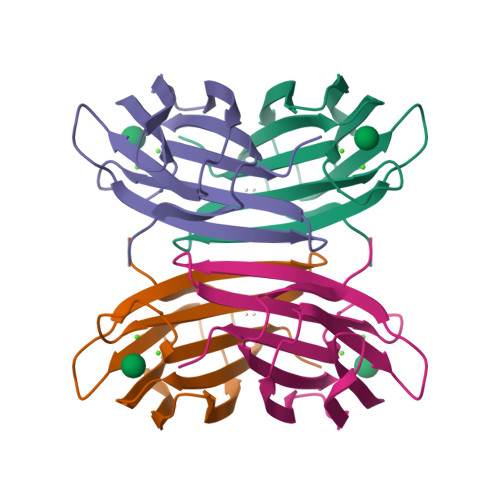
A new Ralstonia solanacearum high-affinity mannose-binding lectin RS-IIL structurally resembling the Pseudomonas aeruginosa fucose-specific lectin PA-IIL.
Sudakevitz, D., Kostlanova, N., Blatman-Jan, G., Mitchell, E.P., Lerrer, B., Wimmerova, M., Katcoff, D.J., Imberty, A., Gilboa-Garber, N.(2004) Mol Microbiol 52: 691-700
- PubMed: 15101976
- DOI: https://doi.org/10.1111/j.1365-2958.2004.04020.x
- Primary Citation of Related Structures:
1UQX, 2CHH - PubMed Abstract:
The plant pathogen Ralstonia solanacearum produces two lectins, each with different affinity to fucose. We described previously the properties and sequence of the first lectin, RSL (subunit M(r) 9.9 kDa), which is related to fungal lectins (Sudakevitz, D., Imberty, A., and Gilboa-Garber, N., 2002, J Biochem 132: 353-358). The present communication reports the discovery of the second one, RS-IIL (subunit M(r) 11.6 kDa), a tetrameric lectin, with high sequence similarity to the fucose-binding lectin PA-IIL of Pseudomonas aeruginosa. RS-IIL recognizes fucose but displays much higher affinity to mannose and fructose, which is opposite to the preference spectrum of PA-IIL. Determination of the crystal structure of RS-IIL complexed with a mannose derivative demonstrates a tetrameric structure very similar to the recently solved PA-IIL structure (Mitchell, E., et al., 2002, Nature Struct Biol 9: 918-921). Each monomer contains two close calcium cations that mediate the binding of the monosaccharide and explain the outstandingly high affinity to the monosaccharide ligand. The binding loop of the cations is fully conserved in RS-IIL and PA-IIL, whereas the preference for mannose versus fucose can be attributed to the change of a three-amino-acid sequence in the 'specificity loop'.
Organizational Affiliation:
Faculty of Life Sciences, Bar-Ilan University, Ramat-Gan, 52900, Israel.