Structure-Based Design of Novel HIV-1 Protease Inhibitors To Combat Drug Resistance.
Ghosh, A.K., Sridhar, P.R., Leshchenko, S., Hussain, A.K., Li, J., Kovalevsky, A.Y., Walters, D.E., Wedekind, J.E., Grum-Tokars, V., Das, D., Koh, Y., Maeda, K., Gatanaga, H., Weber, I.T., Mitsuya, H.(2006) J Med Chem 49: 5252-5261
- PubMed: 16913714
- DOI: https://doi.org/10.1021/jm060561m
- Primary Citation of Related Structures:
2HB3 - PubMed Abstract:
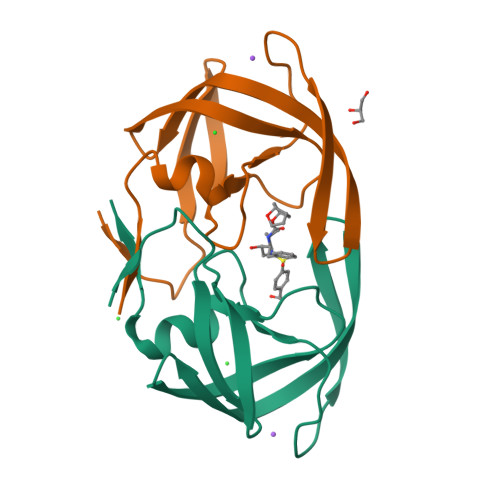
Structure-based design and synthesis of novel HIV protease inhibitors are described. The inhibitors are designed specifically to interact with the backbone of HIV protease active site to combat drug resistance. Inhibitor 3 has exhibited exceedingly potent enzyme inhibitory and antiviral potency. Furthermore, this inhibitor maintains impressive potency against a wide spectrum of HIV including a variety of multi-PI-resistant clinical strains. The inhibitors incorporated a stereochemically defined 5-hexahydrocyclopenta[b]furanyl urethane as the P2-ligand into the (R)-(hydroxyethylamino)sulfonamide isostere. Optically active (3aS,5R,6aR)-5-hydroxy-hexahydrocyclopenta[b]furan was prepared by an enzymatic asymmetrization of meso-diacetate with acetyl cholinesterase, radical cyclization, and Lewis acid-catalyzed anomeric reduction as the key steps. A protein-ligand X-ray crystal structure of inhibitor 3-bound HIV-1 protease (1.35 A resolution) revealed extensive interactions in the HIV protease active site including strong hydrogen bonding interactions with the backbone. This design strategy may lead to novel inhibitors that can combat drug resistance.
Organizational Affiliation:
Department of Chemistry and Medicinal Chemistry, Purdue University, West Lafayette, Indiana 47907, USA. akghosh@purdue.edu