Evidence for Heme Oxygenase Activity in a Heme Peroxidase.
Raven, E.L., Badyal, S.K., Eaton, G., Mistry, S., Pipirou, Z., Basran, J., Metcalfe, C.L., Gumiero, A., Handa, S., Moody, P.C.E.(2009) Biochemistry 48: 4738
- PubMed: 19309109
- DOI: https://doi.org/10.1021/bi900118j
- Primary Citation of Related Structures:
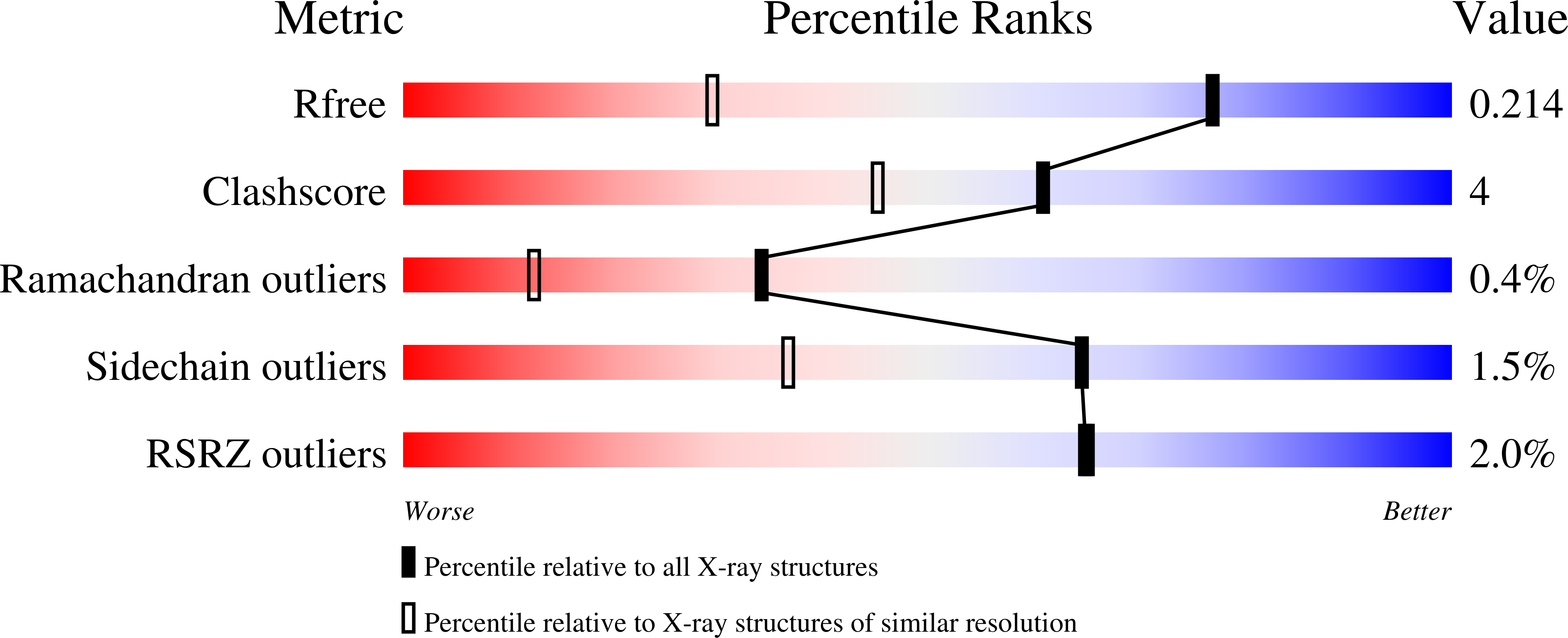
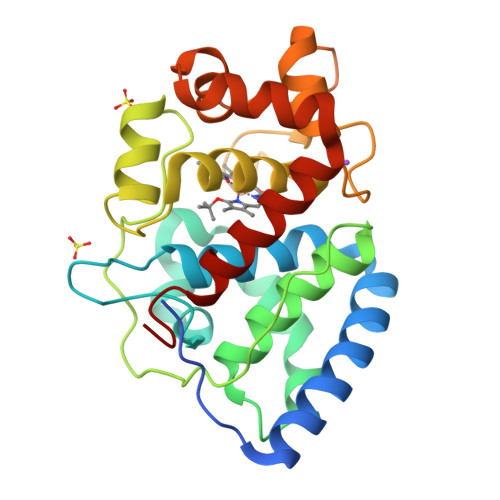
2WD4 - PubMed Abstract:
The heme peroxidase and heme oxygenase enzymes share a common heme prosthetic group but catalyze fundamentally different reactions, the first being H(2)O(2)-dependent oxidation of substrate using an oxidized Compound I intermediate, and the second O(2)-dependent degradation of heme. It has been proposed that these enzymes utilize a common reaction intermediate, a ferric hydroperoxide species, that sits at a crossroads in the mechanism and beyond which there are two mutually exclusive mechanistic pathways. Here, we present evidence to support this proposal in a heme peroxidase. Hence, we describe kinetic data for a variant of ascorbate peroxidase (W41A) which reacts slowly with tert-butyl hydroperoxide and does not form the usual peroxidase Compound I intermediate; instead, structural data show that a product is formed in which the heme has been cleaved at the alpha-meso position, analogous to the heme oxygenase mechanism. We interpret this to mean that the Compound I (peroxidase) pathway is shut down, so that instead the reaction intermediate diverts through the alternative (heme oxygenase) route. A mechanism for formation of the product is proposed and discussed in the light of what is known about the heme oxygenase reaction mechanism.
Organizational Affiliation:
Department of Chemistry, Henry Wellcome Building, University of Leicester, University Road, Leicester LE1 7RH, UK.