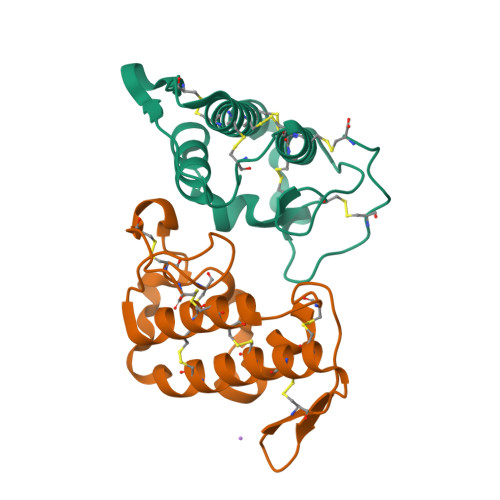
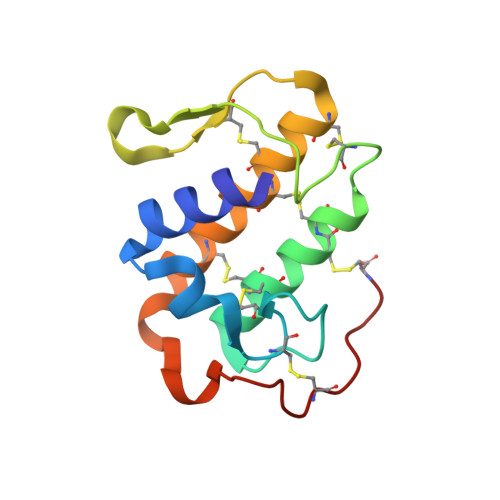
Comparison between apo and complexed structures of bothropstoxin-I reveals the role of Lys122 and Ca(2+)-binding loop region for the catalytically inactive Lys49-PLA(2)s.
Fernandes, C.A., Marchi-Salvador, D.P., Salvador, G.M., Silva, M.C., Costa, T.R., Soares, A.M., Fontes, M.R.(2010) J Struct Biol 171: 31-43
- PubMed: 20371382
- DOI: https://doi.org/10.1016/j.jsb.2010.03.019
- Primary Citation of Related Structures:
3HZD, 3HZW, 3I03, 3I3I, 3IQ3 - PubMed Abstract:
Phospholipases A(2) (Asp49-PLA(2)s) are enzymes responsible for cellular membrane disruption through Ca(2+)-dependent hydrolysis of phospholipids. A class of these proteins (Lys49-PLA(2)s) does not show catalytic activity but can exert a pronounced local myotoxic effect that is not neutralized by serum therapy. In this work, we present five structures of Lys49-PLA(2)s from snakes of the Bothrops genus in apo form, complexed with PEG molecules and chemically modified by p-bromofenacil bromide (BPB), a classic inhibitor of PLA(2). We present herein an extensive structural analysis including: (i) the function of hydrophobic long-chain molecules as Lys49-PLA(2)s inhibitors, (ii) the role of Lys122, previously indicated as being responsible for Lys49-PLA(2)s catalytic inactivity and, (iii) a structural comparison of the Ca(2+)-binding loop region between Lys49 and Asp49-PLA(2)s. The Lys122 analysis of 30 different monomers for apo and complexed Lys49-PLA(2)s structures shows that this residue is very flexible and may bind to different carboxyl groups giving stability to the crystal structures. The structural comparisons of the Ca(2+)-binding loop region between Lys49 and Asp49-PLA(2)s reveal the importance of the Tyr28 residue conservation in Asp49-PLA(2)s to the integrity of this loop. The Tyr28 residue stabilizes this region by an interaction with Gly35 residue. In Lys49-PLA(2)s and low-catalytic Asp49-PLA(2)s this interaction does not occur, preventing the binding of Ca(2+).
Organizational Affiliation:
Departamento de Física e Biofísica, Instituto de Biociências, UNESP - Univ Estadual Paulista, Botucatu, SP, Brazil.