Design, Synthesis, and Investigation of Novel Nitric Oxide (NO)-Releasing Prodrugs as Drug Candidates for the Treatment of Ischemic Disorders: Insights into NO-Releasing Prodrug Biotransformation and Hemoglobin-NO Biochemistry.
Xu, G.G., Deshpande, T.M., Ghatge, M.S., Mehta, A.Y., Omar, A.S., Ahmed, M.H., Venitz, J., Abdulmalik, O., Zhang, Y., Safo, M.K.(2015) Biochemistry 54: 7178-7192
- PubMed: 26582149
- DOI: https://doi.org/10.1021/acs.biochem.5b01074
- Primary Citation of Related Structures:
5E29 - PubMed Abstract:
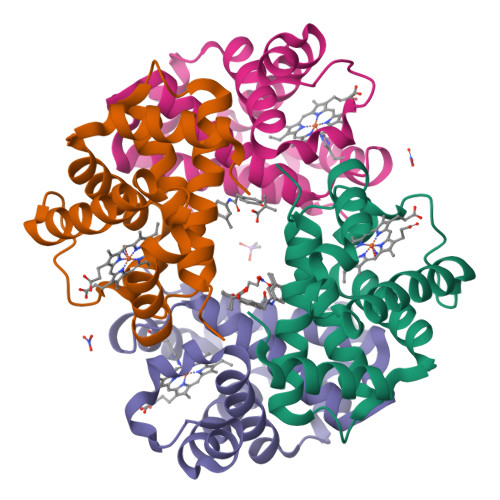
We have developed novel nitric oxide (NO)-releasing prodrugs of efaproxiral (RSR13) for their potential therapeutic applications in a variety of diseases with underlying ischemia. RSR13 is an allosteric effector of hemoglobin (Hb) that decreases the protein's affinity for oxygen, thereby increasing tissue oxygenation. NO, because of its vasodilatory property, in the form of ester prodrugs has been found to be useful in managing several cardiovascular diseases by increasing blood flow and oxygenation in ischemic tissues. We synthesized three NO-donor ester derivatives of RSR13 (DD-1, DD-2, and DD-3) by attaching the NO-releasing moieties nitrooxyethyl, nitrooxypropyl, and 1-(pyrrolidin-1-yl)diazen-1-ium-1,2-diolate, respectively, to the carboxylate of RSR13. In vitro studies demonstrated that the compounds released NO in a time-dependent manner upon being incubated with l-cysteine (1.8-9.3%) or human serum (2.3-52.5%) and also reduced the affinity of Hb for oxygen in whole blood (ΔP50 of 4.9-21.7 mmHg vs ΔP50 of 25.4-32.1 mmHg for RSR13). Crystallographic studies showed RSR13, the hydrolysis product of the reaction between DD-1 and deoxygenated Hb, bound to the central water cavity of Hb. Also, the hydrolysis product, NO, was observed exclusively bound to the two α hemes, the first such HbNO structure to be reported, capturing the previously proposed physiological bis-ligated nitrosylHb species. Finally, nitrate was observed bound to βHis97. Ultraperformance liquid chromatography-mass spectrometry analysis of the compounds incubated with matrices used for the various studies demonstrated the presence of the predicted reaction products. Our findings, beyond the potential therapeutic application, provide valuable insights into the biotransformation of NO-releasing prodrugs and their mechanism of action and into hemoglobin-NO biochemistry at the molecular level.
Organizational Affiliation:
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, King Abdulaziz University , Alsulaymanyah, Jeddah 21589, Saudi Arabia.