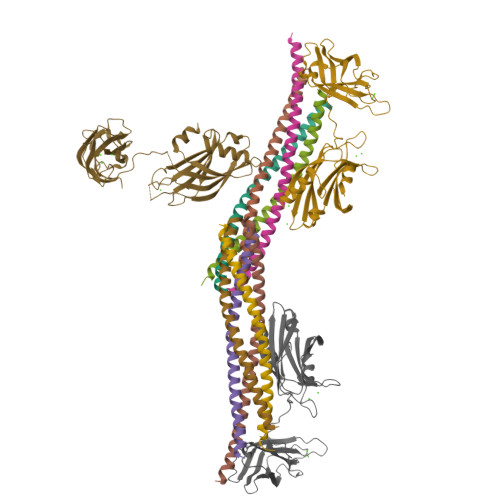
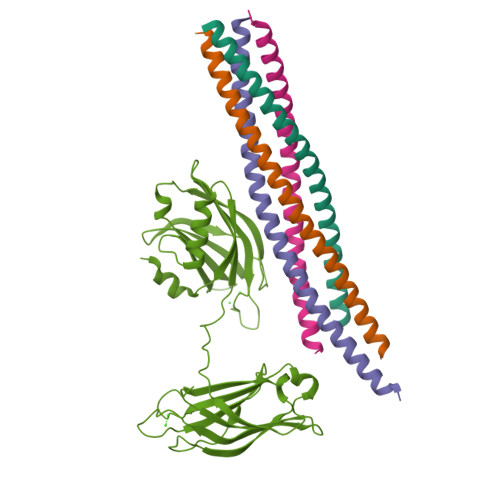
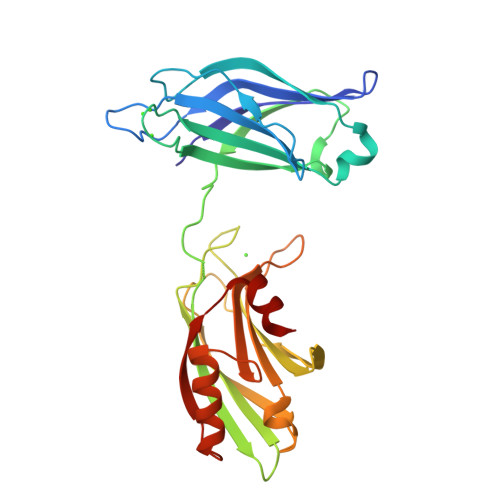
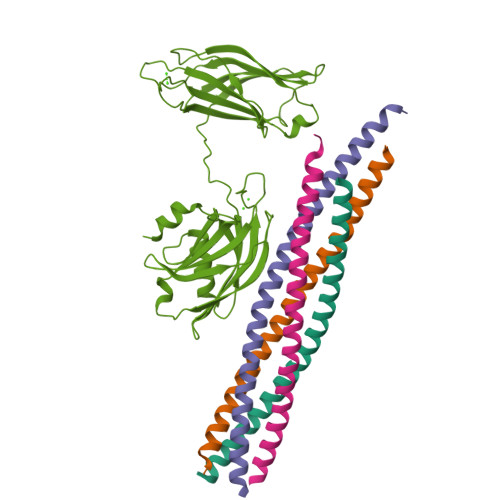
Advances in X-ray free electron laser (XFEL) diffraction data processing applied to the crystal structure of the synaptotagmin-1 / SNARE complex.
Lyubimov, A.Y., Uervirojnangkoorn, M., Zeldin, O.B., Zhou, Q., Zhao, M., Brewster, A.S., Michels-Clark, T., Holton, J.M., Sauter, N.K., Weis, W.I., Brunger, A.T.(2016) Elife 5
- PubMed: 27731796
- DOI: https://doi.org/10.7554/eLife.18740
- Primary Citation of Related Structures:
5KJ7, 5KJ8 - PubMed Abstract:
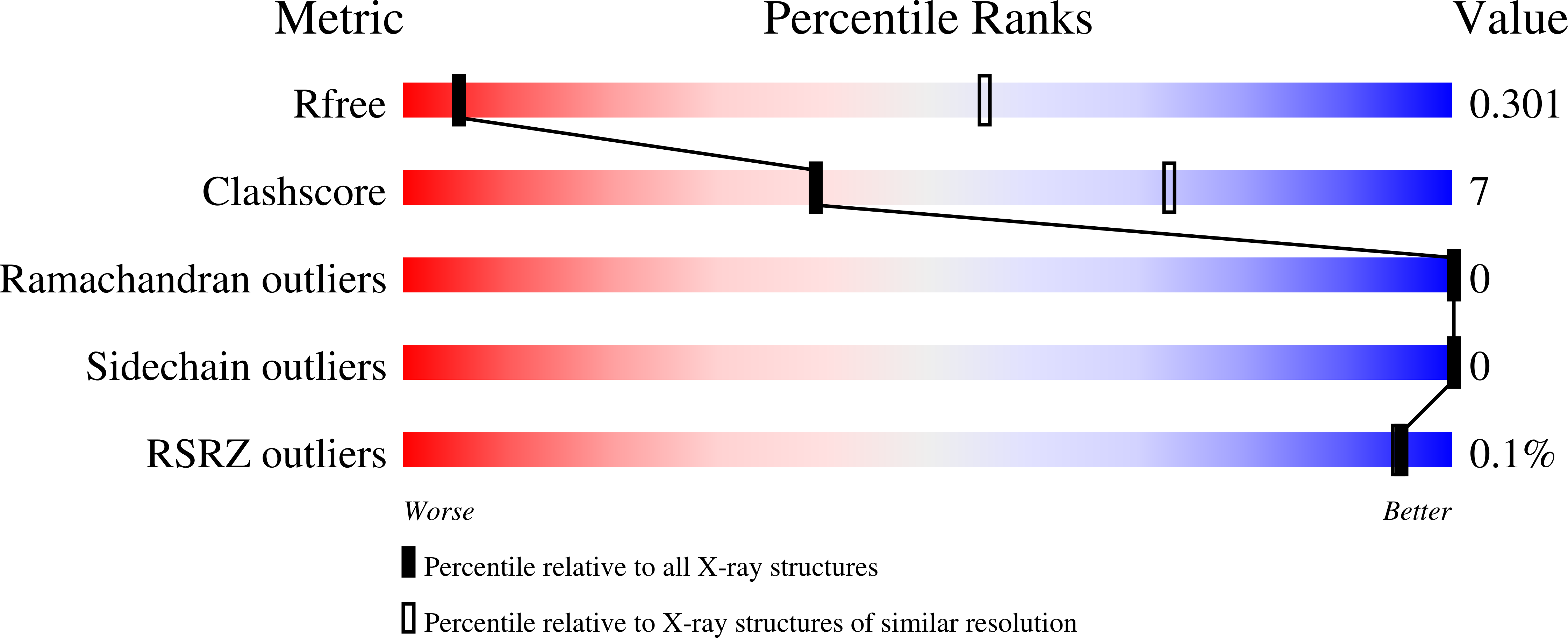
X-ray free electron lasers (XFELs) reduce the effects of radiation damage on macromolecular diffraction data and thereby extend the limiting resolution. Previously, we adapted classical post-refinement techniques to XFEL diffraction data to produce accurate diffraction data sets from a limited number of diffraction images (Uervirojnangkoorn et al., 2015), and went on to use these techniques to obtain a complete data set from crystals of the synaptotagmin-1 / SNARE complex and to determine the structure at 3.5 Å resolution (Zhou et al., 2015). Here, we describe new advances in our methods and present a reprocessed XFEL data set of the synaptotagmin-1 / SNARE complex. The reprocessing produced small improvements in electron density maps and the refined atomic model. The maps also contained more information than those of a lower resolution (4.1 Å) synchrotron data set. Processing a set of simulated XFEL diffraction images revealed that our methods yield accurate data and atomic models.
Organizational Affiliation:
Department of Molecular and Cellular Physiology, Stanford University, Stanford, United States.