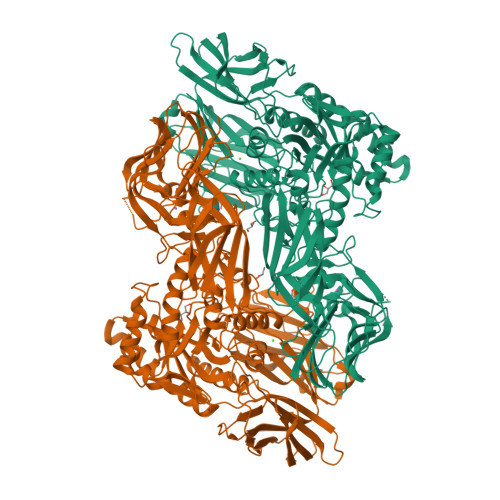
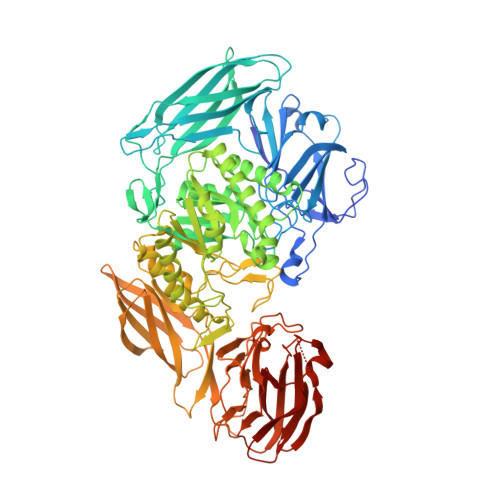
An Atlas of beta-Glucuronidases in the Human Intestinal Microbiome.
Pollet, R.M., D'Agostino, E.H., Walton, W.G., Xu, Y., Little, M.S., Biernat, K.A., Pellock, S.J., Patterson, L.M., Creekmore, B.C., Isenberg, H.N., Bahethi, R.R., Bhatt, A.P., Liu, J., Gharaibeh, R.Z., Redinbo, M.R.(2017) Structure 25: 967-977.e5
- PubMed: 28578872
- DOI: https://doi.org/10.1016/j.str.2017.05.003
- Primary Citation of Related Structures:
5UJ6 - PubMed Abstract:
Microbiome-encoded β-glucuronidase (GUS) enzymes play important roles in human health by metabolizing drugs in the gastrointestinal (GI) tract. The numbers, types, and diversity of these proteins in the human GI microbiome, however, remain undefined. We present an atlas of GUS enzymes comprehensive for the Human Microbiome Project GI database. We identify 3,013 total and 279 unique microbiome-encoded GUS proteins clustered into six unique structural categories. We assign their taxonomy, assess cellular localization, reveal the inter-individual variability within the 139 individuals sampled, and discover 112 novel microbial GUS enzymes. A representative in vitro panel of the most common GUS proteins by read abundances highlights structural and functional variabilities within the family, including their differential processing of smaller glucuronides and larger carbohydrates. These data provide a sequencing-to-molecular roadmap for examining microbiome-encoded enzymes essential to human health.
Organizational Affiliation:
Department of Chemistry, University of North Carolina at Chapel Hill, Chapel Hill, NC 27599, USA.