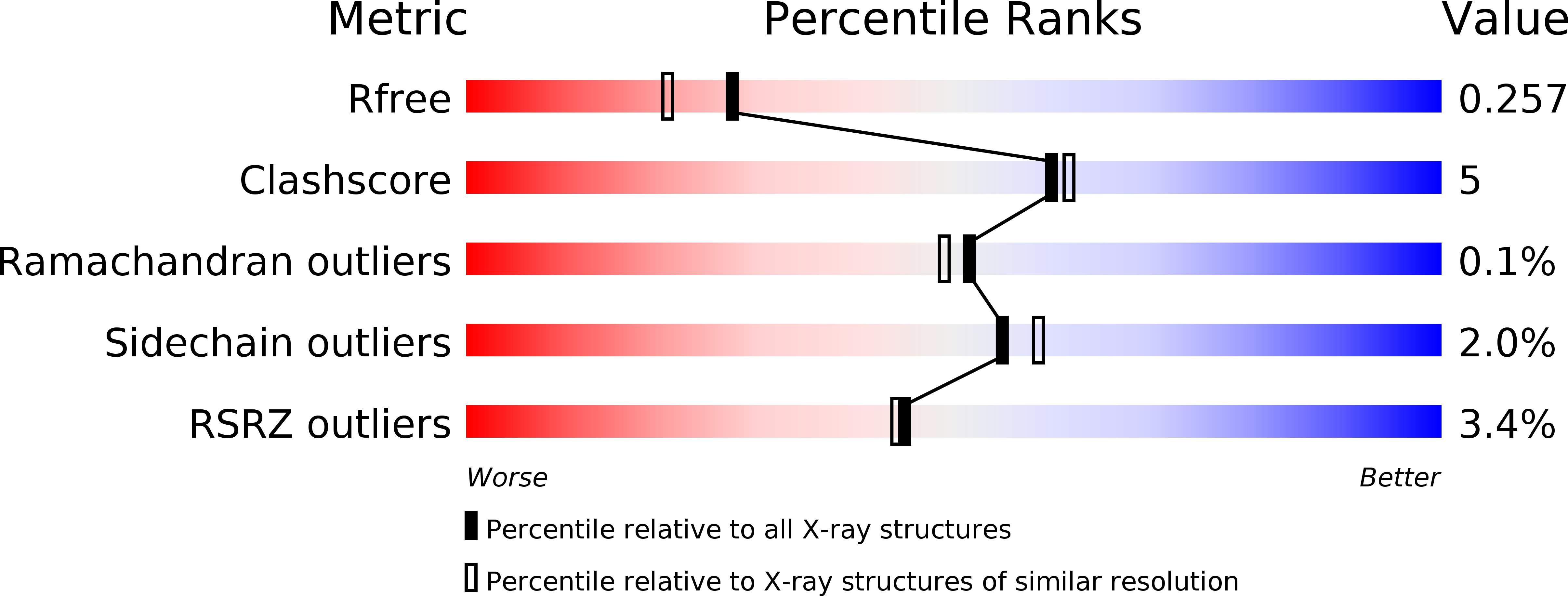
Synthesis and Biological Evaluation of 1,3-Dideazapurine-Like 7-Amino-5-Hydroxymethyl-Benzimidazole Ribonucleoside Analogues as Aminoacyl-tRNA Synthetase Inhibitors.
Zhang, B., Pang, L., Nautiyal, M., De Graef, S., Gadakh, B., Lescrinier, E., Rozenski, J., Strelkov, S.V., Weeks, S.D., Van Aerschot, A.(2020) Molecules 25
- PubMed: 33081246
- DOI: https://doi.org/10.3390/molecules25204751
- Primary Citation of Related Structures:
7AP1, 7AP2, 7AP3, 7AP4 - PubMed Abstract:
Aminoacyl-tRNA synthetases (aaRSs) have become viable targets for the development of antimicrobial agents due to their crucial role in protein translation. A series of six amino acids were coupled to the purine-like 7-amino-5-hydroxymethylbenzimidazole nucleoside analogue following an optimized synthetic pathway. These compounds were designed as aaRS inhibitors and can be considered as 1,3-dideazaadenine analogues carrying a 2-hydroxymethyl substituent. Despite our intentions to obtain N 1 -glycosylated 4-aminobenzimidazole congeners, resembling the natural purine nucleosides glycosylated at the N 9 -position, we obtained the N 3 -glycosylated benzimidazole derivatives as the major products, resembling the respective purine N 7 -glycosylated nucleosides. A series of X-ray crystal structures of class I and II aaRSs in complex with newly synthesized compounds revealed interesting interactions of these "base-flipped" analogues with their targets. While the exocyclic amine of the flipped base mimics the reciprocal interaction of the N 3 -purine atom of aminoacyl-sulfamoyl adenosine (aaSA) congeners, the hydroxymethyl substituent of the flipped base apparently loses part of the standard interactions of the adenine N 1 and the N 6 -amine as seen with aaSA analogues. Upon the evaluation of the inhibitory potency of the newly obtained analogues, nanomolar inhibitory activities were noted for the leucine and isoleucine analogues targeting class I aaRS enzymes, while rather weak inhibitory activity against the corresponding class II aaRSs was observed. This class bias could be further explained by detailed structural analysis.
Organizational Affiliation:
Rega Institute for Medical Research, Medicinal Chemistry, KU Leuven, Herestraat 49-box 1041, 3000 Leuven, Belgium.