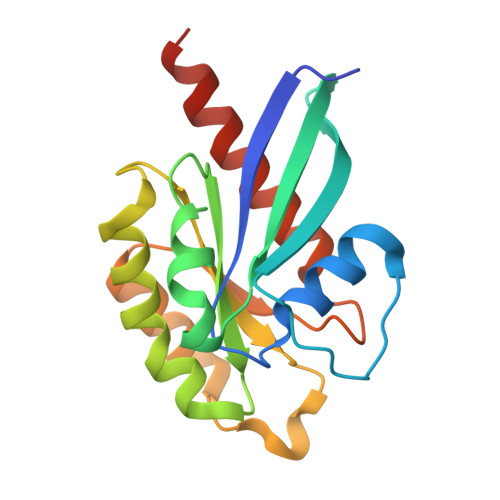
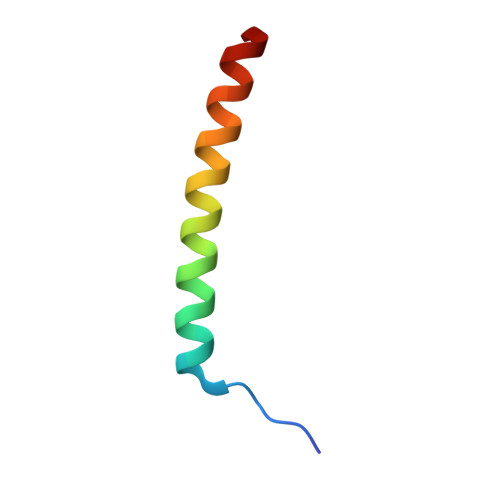
Molecular Basis of the Recognition of the Active Rab8a by Optineurin.
Zhang, J., Liu, L., Li, M., Liu, H., Gong, X., Tang, Y., Zhang, Y., Zhou, X., Lin, Z., Guo, H., Pan, L.(2024) J Mol Biol 436: 168811-168811
- PubMed: 39374890
- DOI: https://doi.org/10.1016/j.jmb.2024.168811
- Primary Citation of Related Structures:
9IKQ - PubMed Abstract:
Optineurin (OPTN), a multifunctional adaptor protein in mammals, plays critical roles in many cellular processes, such as vesicular trafficking and autophagy. Notably, mutations in optineurin are directly associated with many human diseases, such as amyotrophic lateral sclerosis (ALS). OPTN can specifically recognize Rab8a and the GTPase-activating protein TBC1D17, and facilitate the inactivation of Rab8a mediated by TBC1D17, but with poorly understood mechanism. Here, using biochemical and structural approaches, we systematically characterize the interaction between OPTN and Rab8a, revealing that OPTN selectively recognizes the GTP-bound active Rab8a through its leucine-zipper domain (LZD). The determined crystal structure of OPTN LZD in complex with the active Rab8a not only elucidates the detailed binding mechanism of OPTN with Rab8a but also uncovers a unique binding mode of Rab8a with its effectors. Furthermore, we demonstrate that the central coiled-coil domain of OPTN and the active Rab8a can simultaneously interact with the TBC domain of TBC1D17 to form a ternary complex. Finally, based on the OPTN LZD/Rab8a complex structure and relevant biochemical analyses, we also evaluate several known ALS-associated mutations found in the LZD of OPTN. Collectively, our findings provide mechanistic insights into the interaction of OPTN with Rab8a, expanding our understanding of the binding modes of Rab8a with its effectors and the potential etiology of diseases caused by OPTN mutations.
Organizational Affiliation:
College of Chemistry and Materials Science, Sichuan Normal University, Chengdu, Sichuan 610068, China; State Key Laboratory of Chemical Biology, Shanghai Institute of Organic Chemistry, University of Chinese Academy of Sciences, Chinese Academy of Sciences, Shanghai 200032, China.